Some of the greatest scientific innovations have occurred because someone was clever enough not to reinvent the wheel—and instead to think of new ways to use something that already exists in nature.
Jeanne Lawrence is just such a woman. A scientist in pediatrics and developmental biology at the University of Massachusetts Medical School, Lawrence studies chromosomal disorders. These conditions are far more common that one might expect, affecting 1 in 140 live births in the U.S. and half of all miscarriages. The most prevalent of these disorders is Down syndrome, in which affected individuals carry three copies of the twenty-first chromosome instead of the usual two.
That third chromosome makes its presence known physiologically in the classic symptoms of Down syndrome: developmental delays and intellectual disability, among others. Biologically, the problem stems from having too many copies of the genes on this chromosome turned on at the same time (genetics, it would seem, does not adhere to the adage that you can’t have too much of a good thing).
There have been any number of approaches to managing Down syndrome or reducing its symptoms. But Lawrence and her team of scientists took a different tack, borrowing a biological mechanism honed by thousands of years of evolution and creatively applying it to try to nip Down syndrome in the bud.
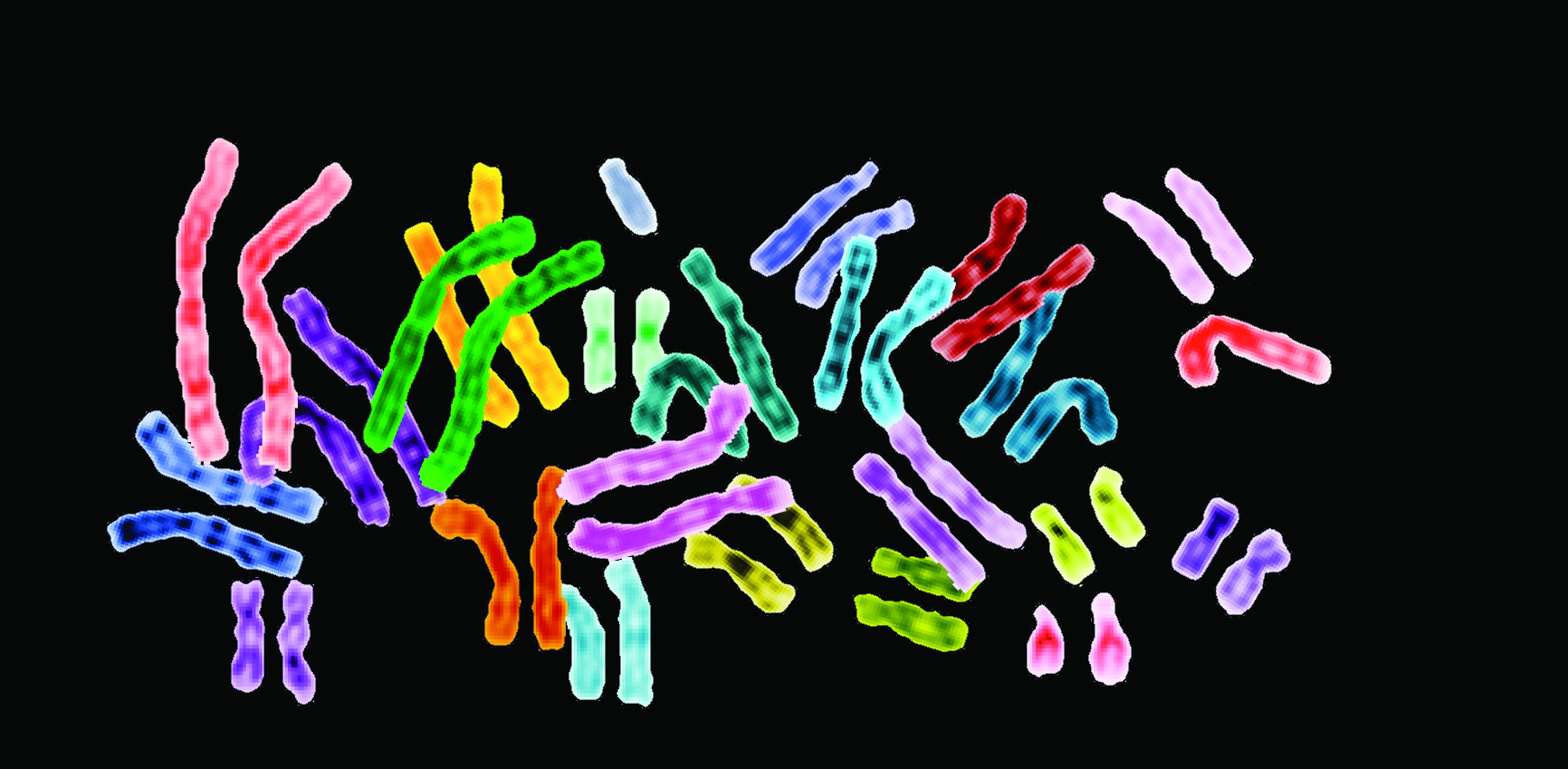
A quick reminder from your high school biology class: while males carry one copy each of the X and Y chromosomes, females have two copies of the X. To prevent females from overdosing on those X-chromosome genes, evolution invented a genetic element whose job is to deactivate half of those X chromosomes. They remain in the genome, but are not functional.
Lawrence, who presented her work at the annual meeting of the American Society of Human Genetics in Boston last week, has come up with a method to scoop that genetic element out of the X chromosome and transfer it, completely intact, to a new home on chromosome 21. Using Down syndrome cells in petri dishes, her team has shown that this approach deactivates the unwanted third chromosome, restoring the treated cells to normal growth patterns and developmental timelines compared to untreated cells. The groundbreaking work has been funded by a special grant for long-shot ideas from the National Institutes of Health. Indeed, the researchers thought the project was so unlikely to succeed that when it did, they validated it in no fewer than eight different ways to be truly certain.
Lawrence is the first to note that this is not a cure for Down syndrome. It can take years to go from tests on cell lines to tests in humans, and at this point the delivery mechanism—or how to insert this remarkable genetic mechanism into the cells of a real person with Down syndrome—remains an enormous and unexplored challenge.
Still, it is extraordinary work that offers a potential that has never been thought possible for people with Down syndrome. Lawrence and her team are moving forward with studies of mice designed to model the condition, hoping that their foray into chromosome therapy will continue to yield promising results.
Hope Seen in Chromosome Therapy for Down Syndrome
There have been any number of approaches to managing Down syndrome or reducing its symptoms. But developmental biologist Jeanne Lawrence and her team at the University of Massachusetts Medical School have taken a different tack, borrowing a biological mechanism honed by thousands of years of evolution and creatively applying it to try to nip Down syndrome in the bud.